Molecular genetic regulation of cytochrome c oxidase (COX) assembly
Aerobic ATP production relies on the coordinate activity of five multimeric protein complexes localized to the inner mitochondrial membrane. One of these, COX, contains two heme and two copper moieties that are required for its catalytic activity. Their synthesis, delivery and insertion into relevant structural subunits during holoenzyme assembly is facilitated by an ever expanding number of proteins termed COX assembly factors. Mutations in many of these ancillary factors have now been identified and cause early onset, fatal diseases with severe, tissue-specific phenotypes that are poorly understood from a clinical perspective.
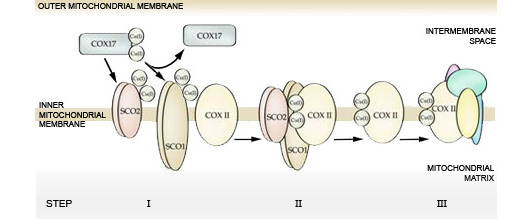
We are studying the molecular function of SCO1 and SCO2, two integral membrane proteins with crucial roles in copper delivery to COX II as it matures and is inserted into the holoenzyme (Jett and Leary, 2018). Using both genetically engineered and patient cell lines, the ultimate goal of this research is to identify and functionally characterize roles of known and novel factors critical to the SCO-dependent metallation of COX II (Jett and Leary, 2018, Boulet et al., 2018, Soma et al., 2019). This will provide for a more complete understanding of the pathways and mechanisms by which copper is delivered to COX during complex biogenesis, and may shed some light as to why two SCO proteins are essential for holoenzyme assembly in some species, while in others only a single SCO protein is required.